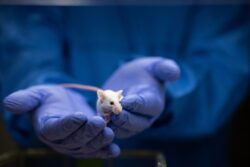
During childhood and adolescence, our brains undergo rapid changes. Studying these changes in juvenile mice has long been challenging because scientists lacked a way to repeatedly image the same animal’s neural pathways as they grow.
Now, researchers at Stanford have developed a revolutionary technique that allows them to do that. By applying a simple solution to a juvenile mouse’s scalp, the team can make the skin transparent to all visible light. This new method creates new opportunities for research on the developing brain that could improve our understanding of neurodevelopmental disorders and lead to new treatment therapies.
‘A literal window’ into brain development
“This opens a literal window to peek into the brain’s development,” said Guosong Hong, an assistant professor of materials science and engineering and senior author on the paper. “Not only can we image the structures of these neurons, but we can also image the neural activity over time in an animal model. In the future, this approach could enable us to look at how these circuits form during the development of an animal.”
The technique is both non-invasive and reversible, meaning scientists can return to the same animal over days and weeks to monitor how its neural circuits evolve. The research, published in PNAS, could advance our understanding of neurodevelopmental disorders and inform new interventions.
Harnessing fundamental laws for new discoveries
Under normal conditions, light scatters when it hits skin. This scattering occurs whenever light waves encounter interfaces between materials with different optical properties, such as lipids, proteins and other molecules within tissue. The effect is similar to trying to see through sunlit fog, blurring what is underneath.
“From a physics perspective, we’re basically a bag of water with biomaterials,” said Mark Brongersma, the Stephen Harris Professor and professor of materials science and engineering and co-author on the paper. “And the mismatch in their optical properties is why we can’t see through the skin or scalp.”
By mixing a compound called ampyrone into water and rubbing it on the mouse’s scalp, the researchers successfully turned the skin transparent.
To overcome this, the team needed to make the water and biomaterials in the skin more similar in their optical properties. This was achieved by raising the refractive index of the water – the degree to which it bends light – to match that of the surrounding biomaterials. By mixing a compound called ampyrone into water and rubbing it on the mouse’s scalp, the researchers successfully turned the skin transparent. Ampyrone absorbs mostly ultraviolet light, allowing scientists to see inside the mouse with the full visible spectrum.
“The fact that such fundamental optics laws can be applied and work in a biological system is just amazing to me,” Brongersma said. “It wasn’t clear whether the physics and the chemistry and the biology would all line up to make this happen.”
Advancing neural imaging
This new method builds on the team’s earlier work, in which they discovered a compound that rendered skin transparent to red light, allowing internal imaging without incisions. Ampyrone goes further, permitting imaging with the full visible spectrum. This makes it possible to observe the colours of green and yellow fluorescent proteins – which are commonly used to mark neural activity.
Because young mice have very thin skulls, fluorescence can be detected until the animals are about four weeks old – the developmental equivalent of a human teenager or early adult. Using this approach, the researchers repeatedly imaged the neurons of sedated juvenile mice and even monitored neural activity in awake mice responding to a puff of air on their whiskers.
This new method provides a non-invasive way to observe brain development in living juvenile mice, offering insights into how neural circuits form and function. By enabling repeated imaging over time, the technique opens new opportunities for studying neurodevelopmental disorders and testing potential interventions. Looking ahead, researchers hope to refine the method and expand its use, ultimately improving our understanding of human brain development and allowing for the production of new treatments for neurological conditions.