Explore how new lab-grown liver organoids can enhance the testing of fibrosis drugs for chronic liver disease treatments.
Researchers at the Institute of Science Tokyo (Science Tokyo) have developed a new lab-grown liver organoid that replicates the regenerative processes of the human liver, offering a new method for testing future treatments of chronic liver disease. The model reproduces interactions between hepatocytes and hepatic stellate cells, two key cell types involved in liver repair and fibrosis. It provides a human-based platform to study liver scarring, cell communication during injury and to test potential drugs that could stop or even reverse liver damage.
Understanding the challenge of liver fibrosis
The human body has an extraordinary ability to heal. However, in chronic liver disease, the repair system can exacerbate harm. Repeated injury triggers cycles of damage and repair, leading to overproduction of extracellular matrix (ECM). As healthy liver tissue is gradually replaced by stiff scar tissue – a process called fibrosis – it can progress to cirrhosis, an irreversible stage often resulting in liver failure. At present, liver transplantation remains the only treatment for advanced cirrhosis, highlighting the urgent need for therapies that can intervene earlier.
A new human liver organoid
To address this, the researchers created a novel human liver organoid, known as iPSC-derived hepatocyte–stellate cell surrounding organoid (iHSO). The organoid was designed to replicate interactions between hepatocytes and hepatic stellate cells (HSCs), which are critical for regulating liver repair.
“This study presents a novel system that helps researchers better understand the interactions between hepatocytes and stellate cells, which may lead to the development of new therapies for various liver diseases,” said Professor Sei Kakinuma of the Department of Clinical and Diagnostic Laboratory Science.
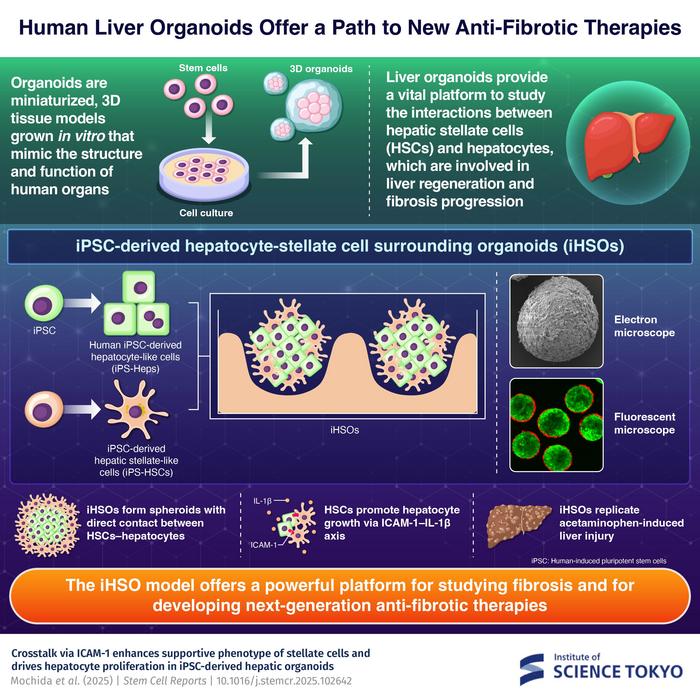
Using human induced pluripotent stem cells, researchers have built a miniature 3D liver model that replicates the critical interaction between hepatocytes and stellate cells. Credit: Institute of Science Tokyo
How the organoid mimics liver repair
In a healthy liver, HSCs remain quiescent, storing vitamin A in small lipid droplets. When injury occurs, they receive signals from damaged hepatocytes as well as from immune and endothelial cells. Activated HSCs transform into myofibroblasts that produce ECM to repair tissue damage.
In a healthy liver, HSCs remain quiescent, storing vitamin A in small lipid droplets.
HSCs and hepatocytes engage in two-way communication: HSCs can promote hepatocyte growth, while hepatocytes influence stellate cell activation or death. Most prior understanding of this relationship comes from animal studies, which do not fully replicate human cell behaviour.
Using human induced pluripotent stem cells (iPS cells), the team generated hepatocyte-like (iPS-Heps) and stellate-like (iPS-HSCs) cells and co-cultured them in three-dimensional organoids, with stellate cells enveloping hepatocytes. The interaction was mediated by ICAM-1, an adhesion molecule, and interleukin-1β (IL-1β), a cytokine produced by HSCs. In iHSOs, iPS-HSCs exhibited a quiescent yet cytokine-rich state, supporting hepatocyte proliferation via the ICAM-1–IL-1β axis.
Modelling liver injury
iHSOs also replicated liver injury. When exposed to acetaminophen – a drug known to cause liver toxicity – the organoids developed injury patterns similar to those seen in human livers, including HSC activation in response to hepatocyte damage.
The iHSO model may one day enable therapies that stop or even reverse liver damage, potentially reducing the need for transplants.
Chronic liver disease affects more than four million adults in the United States, and cases are increasing in Japan due to alcohol consumption and metabolic disorders. By providing a realistic, human-based system, iHSOs could help scientists understand how fibrosis begins and identify drugs that prevent or reverse it.
“iHSO can be applied to liver injury models and is expected to contribute to the elucidation of liver disease pathogenesis and the development of new therapeutic strategies targeting liver fibrosis and regeneration,” says Professor Yasuhiro Asahina.
The iHSO model may one day enable therapies that stop or even reverse liver damage, potentially reducing the need for transplants and providing new treatment options for the millions living with chronic liver disease.